 Exactech and MMI Team Up On Trial (Biloine Young @ OTW)
Exactech and MMI Team Up On Trial (Biloine Young @ OTW)
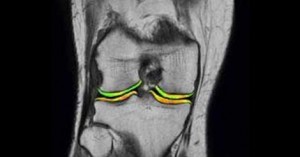
Exactech, which will soon be conducting a BiPhasic Cartilage Repair Implant Clinical Trial that has been approved by the FDA of Taiwan, has engaged Medical Metrics, Inc., (MMI) of Houston, Texas, to evaluate the medical images. The randomized study will be conducted in Taiwan to evaluate a novel cartilage repair treatment.
According to Berkshire Hathaway’s Business Wire, the biphasic implant is a two-phase scaffold that is intended to promote integration with subchondral bone and regeneration of hyaline-like cartilage. During the study, MMI will provide comprehensive analysis and management of MR images.
MMI officials say that the company’s corporate objective is to enter the cartilage repair market and commercialize the new technology in multiple global markets. To do this it developed the imaging sequences and assessment panels necessary to evaluate treatment outcomes, including extent of lesion repair, bone-implant integration, and overall device integrity.
For more than 10 years, MMI has partnered with clinical trial sponsors to conduct comprehensive imaging evaluations of treatments for musculoskeletal, cardiovascular, and neurological disorders. In the April 18 news release, Matt Christensen, Exactech’s Director of Clinical Affairs, said “We chose Medical Metrics for its extensive expertise in musculoskeletal imaging and cartilage repair.”
